Mammalian X Inactivation
| Dosage Compensation | Table of Contents |
Mammalian X inactivation refers to the fact that individuals with more than one X chromosome express the genes on just one of the X chromosomes. The X chromosome being expressed is the active X (Xa) while the other(s) are Xi (inactive). Commonly this occurs in XX individuals so that one copy is inactivated but in cases when an individual has more than two X chromosomes (e.g. XXX) all but one copy is inactivated. Recent studies have also indicated that although we refer to it as X inactivation, it is actually the case that about 20-25% of the genes on Xi are expressed. Therefore, even after X-inactivation XX and XY individuals are not quite the same in terms of their levels of X-linked gene expression.
Inactivation occurs relatively early in development. In each cell, one (or more - from hereon I will just refer to one Xi) X chromosome is chosen at random. The exact process by which one is chosen is still not clear although it appears to involve physical contact between the two X chromosomes! The chosen X is inactivated following the mechanism described below. The result is that it is condensed into heterochromatin and largely unexpressed. The resulting condensed chromosome is referred to as a Barr Body. As development progresses the condensed state is inherited with each cell division so the two daughter cells from a single mitotic division have the same Xi.
One result of this process is that XX individuals can be genetic mosaics. This can occur for any gene for which an individual is heterozygous. Such an individual has the genotype XA1XA2. Since inactivation is random, once it occurs some cells will be expressing the A1 allele while others will be expressing the A2 allele. The result is a mosaic in that different cells are expressing different alleles. (This, of course, will occur for every X-linked heterozygous gene in the individual so there can be a few differences between cells - but of course only two combinations of alleles that can be expressed.)
The result can be (depending on the details of the function of the alleles) variation in phenotype across the cells. Tortoiseshell cats are a classic example of this type of phenotype variation. Since adult cells that are close to one another are often the result of relatively recent cell divisions during development, the inheritance of the Xi state means that local areas of the body will be expressing the same allele.
X inactivation is further described at this link.
The mechanism of inactivation
X inactivation in mammals is an interesting example of both
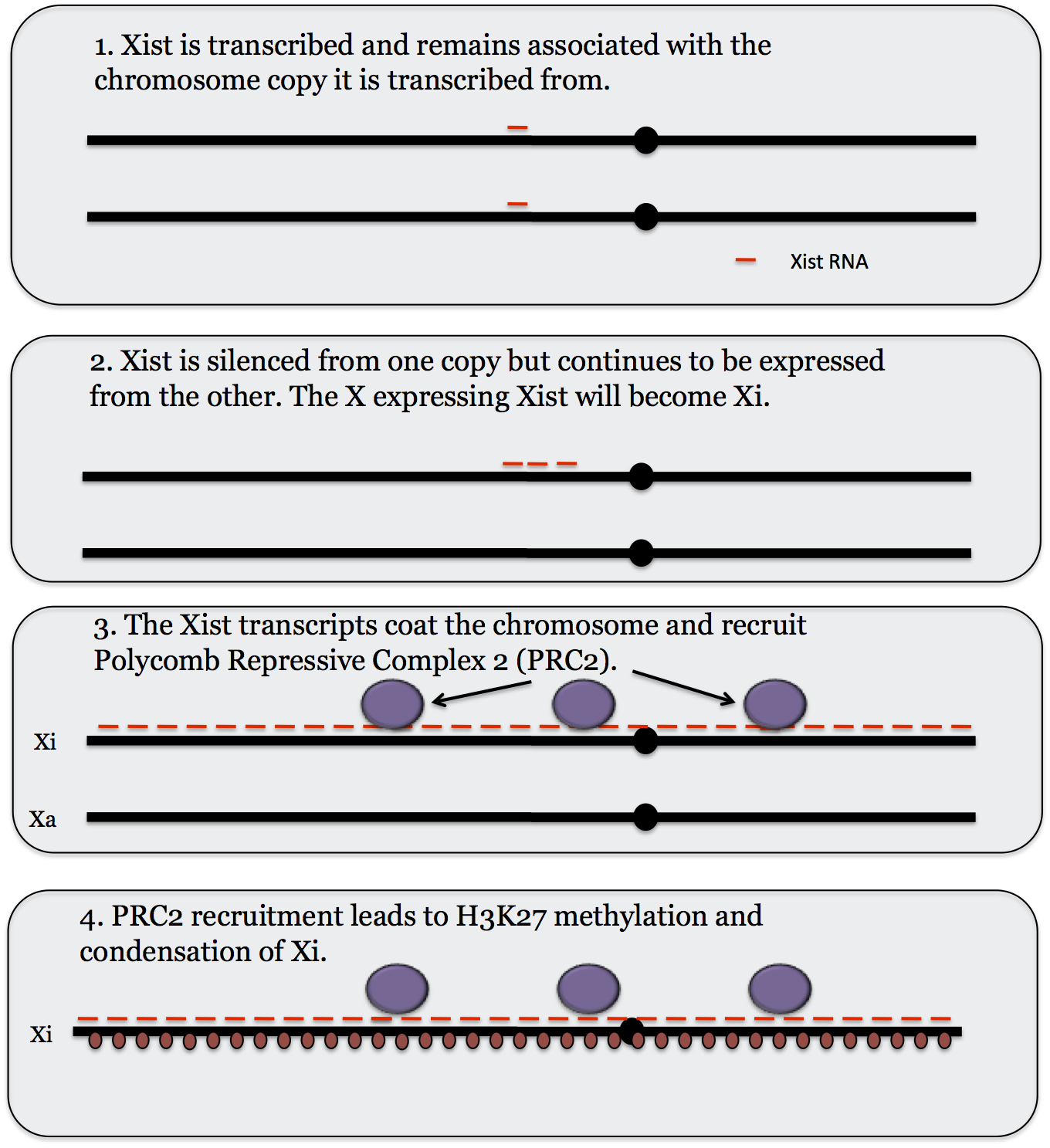
Inactivation of an X chromosome involves the XIC, or X inactivation Center, which is located on the long arm of the X chromosome. This region is now known to code for 4 different lncRNAs; Xist, Tsix, Jpx and Ftx. Here we will only consider Xist which is the one that has been studied the most.
Xist, or X-inactive specific transcript, is expressed from each X chromosome at an early developmental stage. By an unknown process the Xa shuts down expression of Xist while the X to be inactivated increases expression. (Tsix, which codes an RNA that is complementary to Xist, appears to play a role in this.) The Xist RNA product remains associated with the chromosome that expresses it and as it continues to be expressed the additional copies slowly coat the X chromosome, spreading out from XIC. The Xist RNA recruits a Polycomb protein complex called PRC2 (Polycomb Repressive Complex 2). PRC2 in turn induces H3K27 methylation (see the section on histone modification for more on histone methylation) which leads to silencing of most genes on the X chromosome that is coated with Xist.
|
|
The role of Xist gene is also described at Wikipedia.
| Dosage Compensation | Table of Contents |